Osmosis: Permeable to Solute, Impermeable to Solute
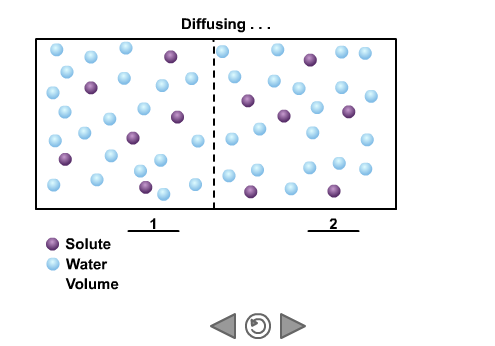 Why does the concentration of a solute cause the movement of water through osmosis? Water is unusual in that it can cross a non-polar membrane but also react and form solutions with ions and polar molecules that cannot cross the same membrane. When water molecules react with a solute they are no longer free to cross the membrane. The more solute, the fewer molecules left that are free to diffuse across the membrane. Therefore a highly concentrated solution (one with lots of solute) has a low effective concentration of water. Therefore water will tend to diffuse across a membrane from areas of low solute concentration to areas of high solute concentration – until the concentration of the solution is equal on both sides. This is only true if the solute cannot cross the membrane.
Why does the concentration of a solute cause the movement of water through osmosis? Water is unusual in that it can cross a non-polar membrane but also react and form solutions with ions and polar molecules that cannot cross the same membrane. When water molecules react with a solute they are no longer free to cross the membrane. The more solute, the fewer molecules left that are free to diffuse across the membrane. Therefore a highly concentrated solution (one with lots of solute) has a low effective concentration of water. Therefore water will tend to diffuse across a membrane from areas of low solute concentration to areas of high solute concentration – until the concentration of the solution is equal on both sides. This is only true if the solute cannot cross the membrane.
Categories and tags of the game : Educational, Science, Single Player
💡 Dato Tecnológico
El uso de Inteligencia Artificial (IA) en el software moderno está revolucionando la experiencia de usuario. Las soluciones de Machine Learning se integran cada vez más en plataformas de entretenimiento.